RINVOQ® (upadacitinib) is indicated for the treatment of adult patients with moderately to severely active ulcerative colitis (UC) who have had an inadequate response, lost response or were intolerant to either conventional therapy or a biologic agent.1
RINVOQ achieved the primary endpoints of clinical remission per adapted Mayo score at Induction Week 8 and Maintenance Week 521,2
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
A Phase 3 trial program involving 3 studies: 2 replicate induction studies (U-ACHIEVE Induction and U-ACCOMPLISH) and 1 maintenance study (U-ACHIEVE Maintenance). A total of 988 patients with moderately to severely active UC evaluating RINVOQ 45 mg QD vs placebo for induction and RINVOQ 15 mg QD and 30 mg QD vs placebo for maintenance treatment (N=451).1*
*Patients who achieved clinical response per adapted Mayo score with 8-week RINVOQ 45 mg QD induction treatment entered maintenance.
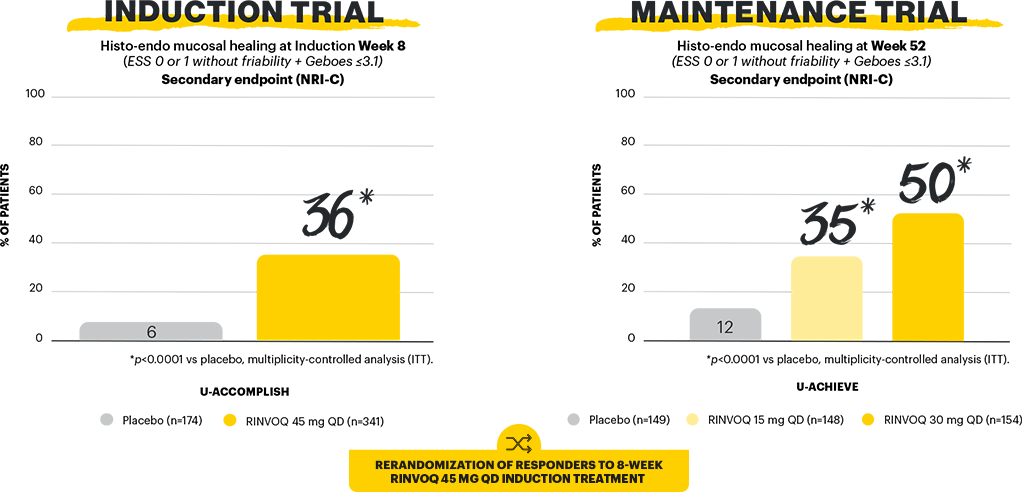
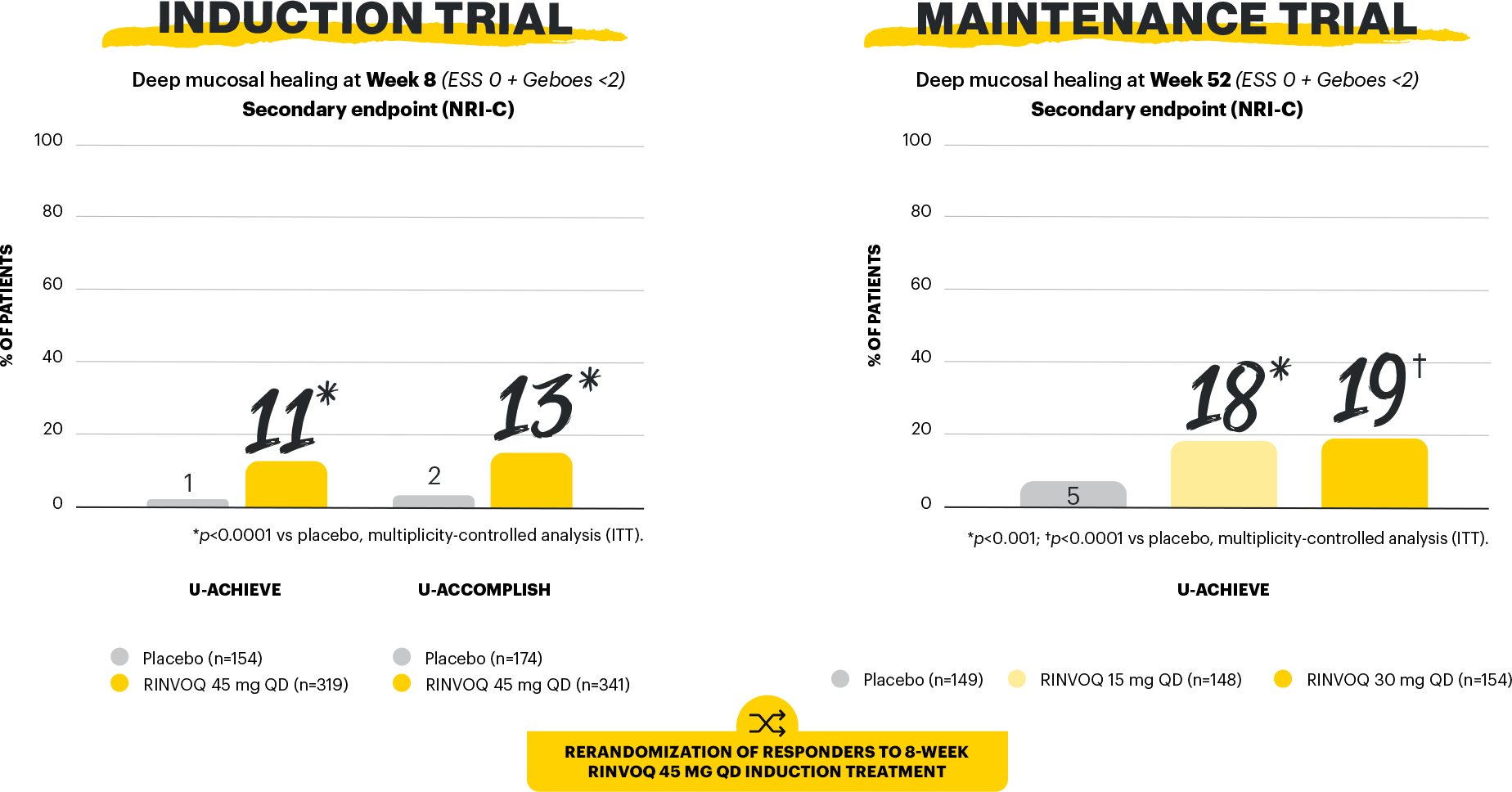
Mucosal healing is a STRIDE-II long-term target of UC treatment.3 In a Phase 3 clinical trial program evaluating the efficacy and safety of RINVOQ in UC, 4 different endpoints were used to measure mucosal healing: mucosal healing, endoscopic remission, histologic-endoscopic mucosal healing, and deep mucosal healing.1,2
aMs: adapted Mayo score; ESS: endoscopic subscore; ITT: intention to treat; NRI-C: non-responder imputation incorporating multiple imputations to handle missing data due to coronavirus disease 2019 (COVID-19); QD: once daily; RBS: rectal bleeding score; STRIDE: Selecting Therapeutic Targets in Inflammatory Bowel Disease; UC: ulcerative colitis.
Study designs: U-ACHIEVE Induction (UC-1) and U-ACCOMPLISH (UC-2) were replicate induction studies, both of which were multicenter, double-blind, placebo-controlled clinical studies. In UC-1 and UC-2, 988 patients (473 and 515 patients, respectively) were randomized to RINVOQ 45 mg QD or placebo for 8 weeks with a 2:1 treatment allocation ratio and included in the efficacy analysis. All enrolled patients had moderately to severely active UC defined as aMs of 5 to 9 with an ESS of 2 or 3 and demonstrated prior treatment failure including inadequate response, loss of response, or intolerance to prior conventional and/or biologic treatment. U-ACHIEVE Maintenance (UC-3) was a multicenter, double-blind, placebo-controlled clinical study with 451 patients who achieved clinical response per aMs (decrease ≥2 points and ≥30% from baseline and a decrease in RBS ≥1 from baseline or an absolute RBS ≤1) with 8-week RINVOQ 45 mg QD induction treatment. Patients were rerandomized 1:1:1 to receive either RINVOQ 15 mg QD, 30 mg QD, or placebo.1,2
Mucosal healing (secondary, multiplicity-controlled endpoint): ESS of 0 or 1 without friability. Maintenance of mucosal healing at Maintenance Week 52 (secondary, multiplicity-controlled endpoint): ESS of 0 or 1 without friability at Maintenance Week 52 in patients who achieved mucosal healing with 8-week RINVOQ 45 mg induction treatment (n=216). Endoscopic remission: ESS=0. Histo-endo mucosal healing (secondary, multiplicity-controlled endpoint): ESS of 0 or 1 without friability and Geboes score of ≤3.1. Deep mucosal healing (secondary, multiplicity-controlled endpoint): ESS of 0 and Geboes score of <2.1,2
UP NEXT
[Please insert local summary of safety]
REFERENCES
- RINVOQ [Summary of Product Characteristics]. AbbVie Deutschland GmbH & Co. KG; December 2023.
- Danese S, Vermeire S, Zhou W, et al. Upadacitinib as induction and maintenance therapy for moderately to severely active ulcerative colitis: results from three phase 3, multicentre, double-blind, randomised trials. Lancet. 2022;399(10341):2113-2128. doi:10.1016/S0140-6736(22)00581-5
- Turner D, Ricciuto A, Lewis A, et al. STRIDE-II: an update on the selecting therapeutic targets in inflammatory bowel disease (STRIDE) initiative of the International Organization for the Study of IBD (IOIBD): determining therapeutic goals for treat-to-target strategies in IBD. Gastroenterology. 2021;160(5):1570-1583. doi:10.1053/j.gastro.2020.12.031